Riccia, a genus of thallose liverworts in the order Marchantiales, is widely distributed both in tropical and temperate regions of the world.
- It belongs to the family of Ricciaceae.
- The plant body of Riccia grows predominantly on damp soil and moist, shady rocks, particularly during and after rain.
| Division: | Bryophyta |
| Class: | Hepaticopsida |
| Order: | Marchantiales |
| Family: | Ricciaceae |
| Genus: | Riccia |
Distribution of Riccia
- Riccia is the most widely distributed genus in the family of Ricciaceae. It is represented by about 138 species.
- Riccia is present throughout all parts of the world.
- They can be found mostly in both tropical and temperate regions.
- Most of the species are terrestrial and prefer to grow on damp soil or moist and shady rocks, especially during and after rain.
- A few are free-floating or submerged aquatic species, e.g., R. fluitans and R. natans. Riccia fluitans occurs floating in still stagnant water or submerged below the surface of standing water.
- Some of the common Riccia species are Riccia discolor, R. frostii, R. crystalline, R. gangetica, R. melanospora, R. hirsuta, etc.
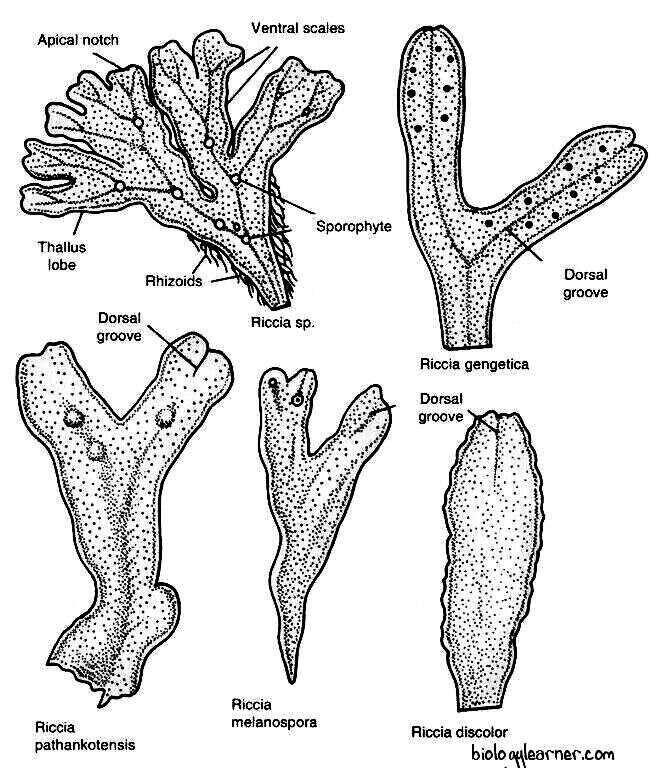
Gametophyte of Riccia
The gametophyte of Riccia is a thallus, which is not differentiated into stems and leaves and lacks true roots. The gametophytic thallus is the dominant phase of the life cycle.
Morphology of Riccia
The plant body of Riccia is gametophytic. The gametophyte is a flat, dichotomously branched, dorsiventral prostrate, ribbon-like, green, fleshy thallus. Each branch of the thallus is linear to wedge-shaped or obcordate.

In terrestrial habits, the plants usually occur in a rosette form or a circular patch due to the presence of a number of dichotomous branches that grow together in one place.
The thallus is 5-7 mm long and 1.2 mm wide in all terrestrial species, while in Riccia fluitans, it is 30-50 mm long and 1-2 mm broad.
Dorsal Surface
The dorsal surface of the Riccia thallus is usually green in color, each branch having a thick midrib in the sagittal axis.
The midrib is traversed by a conspicuous median longitudinal furrow or groove which ends in a depression at the apical region of the thallus, forming an apical notch. The growing point is situated at the tip of the notch.
The main function of the mid-dorsal furrow is to retain the water required for fertilization.
Ventral Surface
The ventral surface of the Riccia thallus bears numerous scales and rhizoids.
Scales or Hairs
Scales are multicellular, one-cell thick, and violet-colored structures that increase the surface area for absorption. The color of the scale is due to the pigment dissolving in the cell sap. The scales are more crowded near the apex and overlap each other at the growing point.
Each scale splits up into two in the mature portion of the thallus and forms two rows of scales along the two margins of the thallus.
Scales are ephemeral (short-lived) in hygrophilous species but persistent and leafy in xerophytic species. In Riccia crystallina, they are inconspicuous and absent.
Rhizoids
The rhizoids are unicellular, elongated, unbranched, tubular hair-like structures present on the ventral surface of the thallus. They appear as prolongations of the lower epidermal cells.
Mature rhizoids are devoid of protoplasm and are absent in aquatic forms (e.g., Riccia fluitans, R. natans).
The main functions of rhizoids are to attach the thallus to the substratum and to absorb water and mineral nutrients from the soil. They are analogous to the roots of higher plants.
Rhizoids are of two types: smooth-walled rhizoids and tuberculate rhizoids.
- Smooth-walled rhizoids: The smooth-walled rhizoids are simple, elongated, and slightly wider. They contain a smooth inner wall with colorless contents.
- Tuberculate or pegged rhizoids: Tuberculate rhizoids are thinner and have peg-like or plate-like projections in the cell lumen.
Anatomy of Riccia
In a vertical cross-section, the thallus shows an internal differentiation of tissues distinctly organized into two horizontal regions, an upper photosynthetic or assimilatory region and a lower storage region.
Photosynthetic Region
The upper dorsal region, i. e., the photosynthetic region, is composed of chlorophyll-bearing cells arranged in vertical rows. The chlorophyllous cells are separated by narrow longitudinal vertical air canals called air chambers.
Each air chamber is generally surrounded by four vertical rows of cells (e.g., Riccia glauca), sometimes eight rows are also present (e.g, R. vesiculosa).
The air chamber communicates with the external atmosphere through an air pore. Each air pore is a simple intracellular space bounded by 4 to 8 colorless enlarged terminal cells of the vertical rows which form a loose discontinuous one-celled thick upper epidermis.
Air pores help in the gaseous exchange.
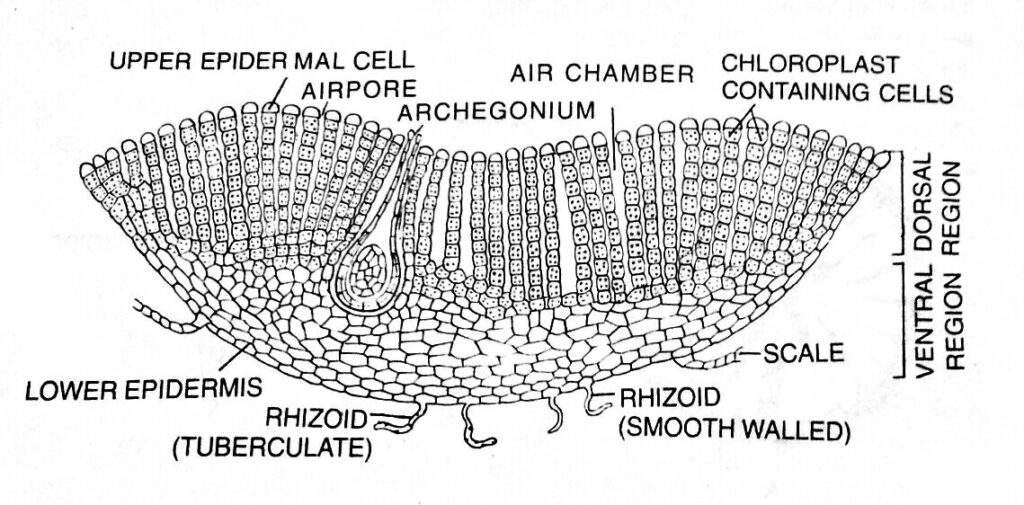
Storage Region
The lower storage region of the thallus is composed of colorless, compactly arranged parenchymatous cells and lies below the photosynthetic region.
The cells of the storage region (parenchymatous cells) are thin-walled, without intercellular spaces. These parenchymatous cells generally lack chlorophyll and contain starch granules as reserve food.
The lower surface of the tissue contains small cells that are compactly arranged, forming a single layer of lower epidermis. Some cells of the lower epidermis extend to form the multicellular, one-celled thick scales and the unicellular tubular and smooth rhizoids.
Reproduction in Riccia
Riccia begins to reproduce when it reaches a certain stage of maturity. Riccia reproduces by both vegetative and sexual methods.
Vegetative Reproduction
Vegetative reproduction in Riccia occurs by the following methods:
Fragmentation
Fragmentation is the most common method of vegetative reproduction in Riccia. In this method, the progressive death and decay of the older part of the thallus (due to aging or drought) from the posterior end reaches dichotomy, the young apical branches get separated. Each such branch grows independently by apical growth and develops into a new thallus.
Adventitious Branches
In some species, such as Riccia fluitans, special adventitious branches (similar to the parent thallus) arise from the ventral surface of the thallus in the midrib region. These branches get separated from the parent thallus, which grows into new thalli.
Persistent Apices
The apex of the thallus in some species (e.g., R. discolor) grows down into the soil due to a prolonged dry summer or towards the end of the growing season. Under favorable conditions, the apices come up and develop into new thalli.
Tubers
The apices of the thallus lobes get thickened to form perennating tubers at the end of the growing season. The perennating tuber can easily survive a period of drought and germinate into a new thallus in a favorable season, e.g., R. perennis, R. bulbifera, R. discolor, and R. vesicata.
Gemma-like Body
In some species (e.g., Riccia glauca), the apical region of the young rhizoids divides and re-divides to form a gemma-like body. It contains chloroplasts and can grow into a new thallus.
Sexual Reproduction
Sexual reproduction in Riccia is of the oogamous type. The gametes are produced in highly specialized reproductive organs. The male gametes, or antherozoids, are produced in the antheridium (male reproductive organ), and the female gamete, or egg, is produced in the archegonium (female reproductive organ). The male gametes and the female gametes are structurally and physiologically different.
Riccia is mostly monoecious or homothallic, i.e., antheridia and archegonia are developed on the same thallus (e.g., R. melanospora, R. crystallina, R. gangetica, R. billardieri, R. cruciata, R. pathankotensis, R. glauca, and R. robusta). But some are also dioecious or heterothallic, i.e., antheridia and archegonia are developed on different thalli (e.g., R. discolor, R. sanguinea, R. bischoffii, R. frostii, and R. pearsonii).
Antheridia and archegonia are produced singly. They have developed acropetally in a linear row on the dorsal median furrow. At first, the sex organs are superficial, but at later stages of development, they are enveloped by the outgrowth of tissues. Finally, they are embedded in a cavity formed by the overarching tissues.
These cavities are called antheridial and archegonial chambers and are open on the dorsal surface by the narrow cylindrical canals.
Antheridium
The male reproductive organ of Riccia is the antheridium. It develops singly in the antheridial chamber.
Development of Antheridium
- The antheridium develops from a single superficial antheridial cell of the dorsal longitudinal furrow of the thallus, called the antheridial initial. The initial cell lies 2 or 3 cells back from the apical cell.
- The antheridial initial enlarges and becomes papillate. It divides transversely to form an upper outer cell and a lower basal cell.
- The basal cell undergoes a few divisions and forms the embedded portion of the antheridial stalk.
- The outer cell enlarges and divides by transverse divisions to form a filament of four cells. The upper two cells are primary antheridial cells, and the lower two cells are primary stalk cells.
- Primary stalk cells undergo a few further divisions to form the stalk of the antheridium.
- Primary antheridial cells divide by two successive vertical divisions at right angles to each other, forming two tiers of four cells each.
- Periclinal divisions now appear in both the tiers of four cells, and there is the formation of eight outer sterile jacket initials or wall initials and eight inner fertile primary androgennial cells.
- Jacket initials undergo several anticlinal divisions and form a single layer of the antheridial jacket.
- Primary androgonial cells undergo further divisions (transversely and vertically), resulting in the formation of a mass of small cubical androgonial cells.
- The last generation of the androgonial cells is known as sperm mother cells or spermatocytes (Smith refers to them as the androcyte mother cells).

Spermatogenesis
Spermatogenesis is the process of transformation of androcyte mother cells into antherozoids or sperm. This process is completed within a few minutes.
According to Black (1913), each androcyte mother cell divides by a diagonal mitotic division without the formation of a cell wall to form two triangular cells called androcytes. Both the androcytes remain enclosed in the wall of the androcyte mother cell with one separate wall.

Each androcyte has a dense protoplast containing a distinct large nucleus and a small extra nuclear granule called a blepharoplast. The blepharoplast lies near the periphery of the androcyte protoplast. Thereafter, the androcyte loses its triangular shape and becomes somewhat oval or rounded. Along with this change in the shape of the androcyte is the elongation of the blepharoplast and the nucleus.
The blepharoplast elongates into a thin cord-like structure with a thickened head, and the androcyte becomes coma-shaped. The cord-like blepharoplast occupies the protoplast up to two-thirds of the periphery. Simultaneously, the nucleus also becomes crescent-shaped, and homogeneous, and it also moves to the periphery of the androcyte protoplast. Ultimately, the nucleus comes into contact with the cord-like blepharoplast.
Two large flagella emerge from the conspicuously thickened part of the blepharoplast. The electron microscopy reveals the standard 9+2 fibrillar organization of the flagellum.
Thus, androcytes undergo metamorphosis into antherozoids or sperm. The unused part of the androcyte forms a small vesicle, which may remain attached to the posterior end of the antherozoid.
Structure of Antheridium
The mature antheridium is an elongated structure. It has a globular or pear-shaped body with a short, few-celled stalk. The multicellular stalk attaches the antheridium to the base of the antheridial chamber (deep pit). Each antheridial chamber opens at the dorsal surface of the thallus by a narrow pore called an ostiole.

The antheridium is present singly in the antheridial chamber. A single-layered sterile jacket surrounds the mass of androcytes, which metamorphoses into antherozoids or sperm.
Antherozoids
The antherozoid is a unicellular, uninucleate, biflagellate, and curved structure.
A pair of whiplash flagella are present at the anterior end of the antherozoid. Both flagella are morphologically similar but differ in function. One flagellum serves for propulsion while the other is for rotation and for changes in direction.
Dehiscence of Antheridium
Water helps in the dehiscence of the mature antheridium. The cubical walls inside the antheridium disappear and the antherozoids remain free in the viscous fluid in the antheridial cavity.
Water enters the antheridial chamber through the ostiole. Due to imbibition, the sterile apical cells of the antheridial jacket enlarge by absorbing water. They become softened and ultimately degenerate to form a distal pore.
The antherozoids, along with the viscous mass, come out through the narrow canal (antheridial pore) of the antheridial chamber to the dorsal surface of the thallus.
Archegonium
The female reproductive organ of Riccia is the archegonium. It develops singly in the archegonial chamber.
Development of Archegonium
- Like the antheridium, the archegonium also develops from a single superficial cell in the dorsal longitudinal furrow of the thallus, called the archegonial initial.
- The superficial cell lies close to the apical cell.
- The archegonial initial enlarges in size and becomes papillate. It first divides transversely into a lower basal cell and an upper outer cell.
- The basal cell takes no further divisions and forms the embedded portion of the archegonial stalk.
- The outer cell enlarges and functions as an archegonial mother cell. It divides by three eccentric vertical walls, resulting in the formation of three sterile peripheral initials and a fourth fertile median cell, the primary axial cell.
- The primary axial cell slightly overtops the peripheral initials.
- Each of the three peripheral initials divides by an anticlinal vertical division to form six jacket initials or envelope cells.
- In this way, the primary axial cell gets encircled by jacket initials.
- Six jacket initials divide transversely into six jacket cells of the upper tier and six jacket cells of the lower tier.
- The upper tier is called neck initials, and it divides by repeated transverse divisions to form a tube-like neck.
- The neck of the archegonium is one cell thick.
- The cells of the neck are arranged in six vertical rows, and each row consists of six to nine cells.
- The lower tier is called venter initials, and it is also divided by repeated transverse divisions to form a single layer of the swollen venter.
- Simultaneously, the primary axial cell divides transversely and unequally to form a large lower central cell and a small upper primary cover cell.
- The central cell divides into a lower primary venter cell and an upper primary neck canal cell.
- The primary neck canal cell undergoes several transverse divisions and forms four neck canal cells.
- The primary ventral cell undergoes an unequal transverse division to form a small ventral canal cell and a large egg.
- The upper primary cover cell divides into two successive vertical divisions at right angles to each other, forming four cover cells that develop the mouth of the archegonium.
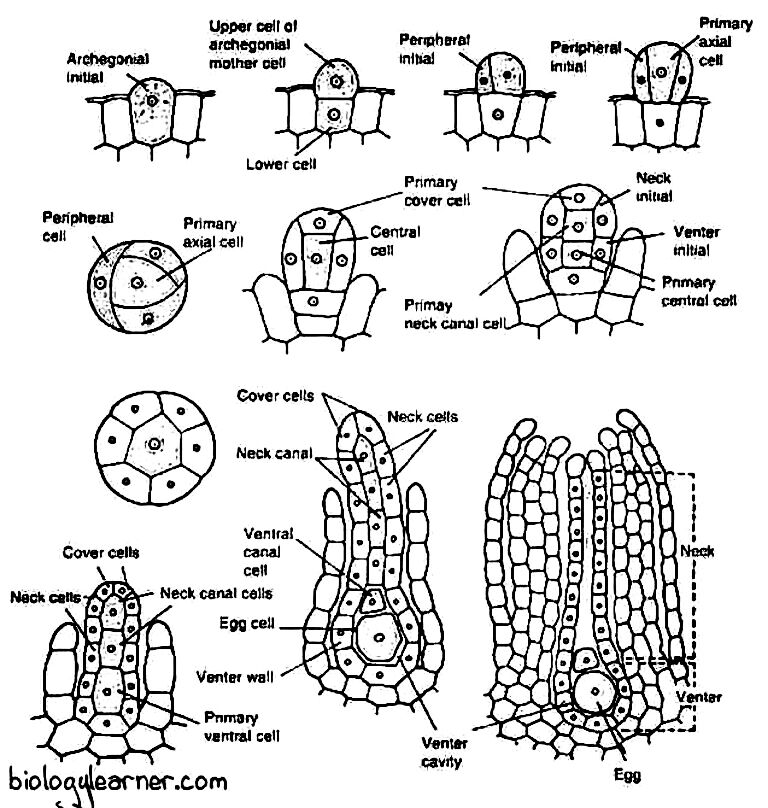
Structure of Archegonium
The mature archegonium is a short-stalked, flask-shaped structure. The stalk attaches it to the thallus.
The archegonium consists of an upper elongated slender neck and a basal swollen portion called the venter.

The long neck is a single-layered tube composed of six vertical rows, and each row consists of six to nine cells. It encircles a narrow canal that consists of a row of four neck canal cells. The apex of the neck is made up of four specialized and slightly large cover cells, or cap cells.
The venter is composed of a single-layered jacket of sterile cells. The jacket encloses a small ventral canal cell and a large naked egg.
Fertilization
Water is necessary for fertilization. The dorsal furrow of the thallus serves as a capillary tube. After rain or heavy dew, water is usually retained in the dorsal furrow in the form of a thin film.
By absorbing water, apical cells of the antheridial wall become swollen. The antheridial wall gets softened and eventually bursts to release a mass of antherozoids.
The liberated antherozoids come up to the dorsal surface of the thallus from the antheridial chamber. They swim in the thin film of water and come near the archegonium due to chemotactic attraction induced by proteins and other chemicals (inorganic salts of potassium) coming out of the hollow archegonial neck.
Meanwhile, in the mature archegonium, the neck canal cells and the single ventral canal cell disintegrate to form a mucilaginous mass. The mucilage imbibes water, swells, and comes out of the archegonial mouth by forcing upon the cover cells, forming a narrow passage called the neck canal.
A number of antherozoids enter the archegonial neck due to the chemotactic response, reach the venter, and ultimately one antherozoid fertilizes the egg to form a diploid zygote or oospore.
After the completion of fertilization, the gametophytic phase ends.
Sporophyte of Riccia
The sporophytic phase is the direct result of the sexual process and begins with the formation of the diploid zygote after fertilization. The zygote or oospore is the first cell of the sporophyte.
After fertilization, the zygote immediately secretes a thin wall around it. It increases in size and almost completely fills the cavity of the venter in the archegonium. Simultaneously, the walls of the venter divide periclinally and then anticlinally to form a two-layered calyptra around the young sporophyte.
Development of Sporophyte
The zygote divides first by a transverse division and forms two almost equal-sized cells. The second division is at a right angle to the first (dividing vertically) and results in the formation of a four-celled embryo, or quadrant stage.
Each quadrant is then divided by another vertical division at a right angle to the preceding one. As a result, the embryo develops into an eight-celled structure or octant stage. The young eight-celled embryo now divides into all possible planes to form a spherical mass of 20–40 undifferentiated colorless cells.

In the peripheral region of the spherical mass, the cells now divide periclinally (parallel with the surface) into an outer layer of amphithecium and an inner mass of cells called endothecium. The cells of the amphithecium divide only by anticlinal walls to form a single-layered protective sterile jacket or capsule wall of the young sporogonium. The endothecium forms the archesporium, which is the first cell generation of sporogenous tissue.
In Riccia crystallina and a few more species, some of the spore mother cells fail to produce spores. Such spore mother cells form abortive nutritive cells called nurse cells. Some bryologists (Pagan, 1932) consider the nurse cells as the forerunners of the elaters, found in more advanced members of the Marchantiales (e.g., Marchantia).
The sporocytes, or spore mother cells, are the last cells of the sporophytic generation, after which it begins to disintegrate. The nurse cells and the amphithecial jacket layer also disintegrate further, followed by the disintegration of the inner cell layers of the ventral wall. All the disintegrated products form a viscous nutritive fluid which serves as nourishment for the spore mother cells and developing spores. The dividing spore mother cells remain suspended in the nutritive fluid.
Sporogenesis
Sporogenesis is the process of the formation of haploid spores from the diploid spore mother cells. The process involves a special kind of nuclear division called meiosis.
The sporogenesis process begins with the contraction of the cytoplasmic contents of the spore mother cells. The spore mother cells now undergo meiosis, dividing by two successive divisions to form haploid spore tetrads.
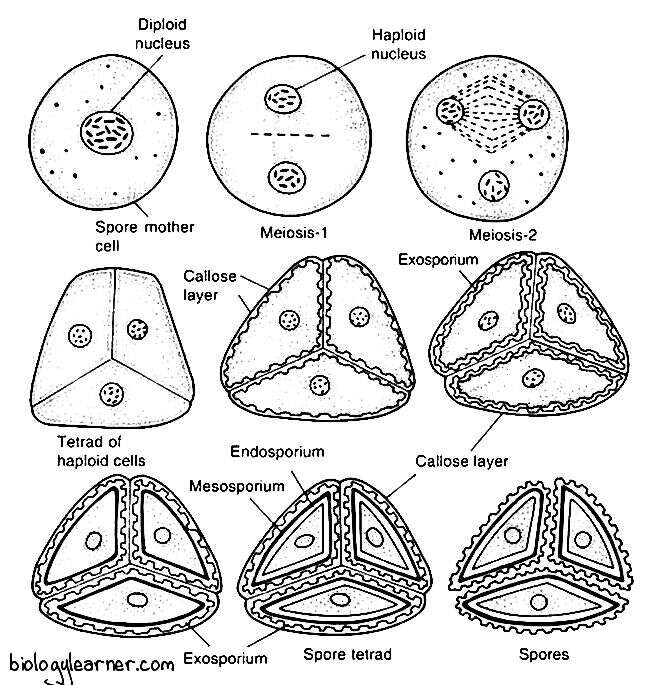
The first meiotic division is the reduction division; thereby, the chromosome number is reduced to half of the somatic number. The cell plate is incomplete during the first meiotic division, and after the completion of the second division, the cell plates delimiting four spores are formed simultaneously.
The spores are tetrahedrally arranged and remain opposite to one another in a spore tetrad. They are also surrounded by the walls of the sporocytes till the maturity of the spore wall. In Riccia personii, four spores are arranged iso-bilaterally.
Structure of Sporophyte
The sporophyte of Riccia is the simplest organization among the liverworts. Both food and seta are completely absent.
The mature sporophyte is represented by only a globular capsule or spore sac. It consists of a single-layered capsule wall (sterile jacket wall) that encloses spores. It also has a bi-layered calyptra which forms a protective covering around the capsule. The calyptra, in fact, is a part of the gametophyte. There are no elaters.

The capsule wall degenerates before the spore mother cells divide to form the spores (spore tetrad) and then the inner layer of the calyptra degenerates. After meiosis, the mass of spores lies free in a cavity or sac surrounded by the outer layer of calyptra. Therefore, the mature sporogonium has no diploid structure (Smith, 1955).
The newly formed spore, the future gametophyte, remains enclosed within the old gametophyte.
Spore
The spores are haploid, uninucleate, and pyramidal in shape. They are very small, 0.05 to 0.12 mm in diameter.
Each spore remains surrounded by three wall layers: the outermost exosporium, the middle mesosporium, and the innermost endosporium.
The outermost exosporium, or perispore, is thin, mucilaginous, ornamented, and very strongly cutinized. The middle-layer mesosporium is differentiated into 3 concentric zones. The innermost endosporium is composed of cellulose and pectose.
Dehiscence of the Capsule and Dispersal of Spores
There is no special method for the dehiscence of the capsule and the dispersal of spores in Riccia.
The jacket layer of the capsule, i. e., sporophyte (sporophyte only consists of the capsule), and the inner layer of the surrounding venter disintegrates long before the spores mature. Although the spore mass remains surrounded by a venter layer inside the gametophytic tissue of the thallus.
In Riccia, the spores are not immediately dispersed after they mature. They remain within the thallus for one year or so.
Ultimately, the spores are set free by the progressive death and decay of the calyptra and the surrounding gametophytic tissues.
Germination of Spores and Formation of the Young Gametophyte
The spore is the first cell of the gametophytic generation.
Each spore germinates in favorable environmental conditions when there is enough water in the soil. Also, light and a low temperature are required for spore germination.
According to Campbell (1918), the exosporium and mesosporium rupture at the triradiate mark, and the endosporium comes out as a tubular germ tube called the germinal. The germinal tube is filled with cytoplasm which contains oil granules, chloroplasts, and albumin granules.
As the content of the cytoplasm moves to the distal end, the germ tube elongates to form a club-shaped structure. The tip portion is now divided by a transverse division to form a small distal cell. It is again divided by a transverse division, which is parallel to the first one.
These two cells undergo two vertical divisions at right angles to one another, forming two tiers of four cells each. This eight-cell structure represents the octant stage.
The distal tier of four cells of the octant functions as an apical cell that divides to produce a new thallus. Along with these divisions, the first rhizoid develops from the base of the germ tube. Many rhizoids develop later on from the new thallus and fix it in the soil.
In Riccia crustissi, the two spores of a spore tetrad develop into male thalli and the other two spores develop into female thalli.
